“A microbially prepared pyrazine diamine compound significantly stabilizes high-energy density lithium-ion batteries, shows a new study by researchers from Japan
Lithium-ion batteries with high-energy-density cathodes are necessary to meet the energy demands of next-generation electronics and electric vehicles. At high voltages, however, the battery electrolyte undergoes excessive decomposition, compromising cathode performance. To tackle this, researchers from Japan have now synthesized a bio-based, non-toxic additive material that stabilizes the cathode by forming a passivation layer on its surface and suppressing its decomposition. Eco-friendly and low-cost, the novel compound could promote a wider utilization of bio-based resources.
High energy density lithium-ion (Li-ion) batteries are indispensable for powering electric and hybrid vehicles, next-generation electronics, and power grids. These Li-ion batteries contain high energy density cathodes based on transition metal oxides. Among numerous investigated potential materials, the LiNi1/3Mn1/3Co1/3O2 cathode has been shown to deliver the best performance at a high potential of 4.5 V versus Li/Li+ with high reversible capacity.
However, at such high potentials, the carbonate species in commercial electrolytes—ethylene carbonate and diethyl carbonate—undergo excessive oxidative decomposition. This, in turn, forms a thick cathode electrolyte interphase (CEI) on the cathode surface, severely compromising its performance. Consequently, researchers have explored electrolyte additives as way to restrict performance degradation by masking and stabilizing the cathode surface. However, currently available options pose safety and environmental hazards.
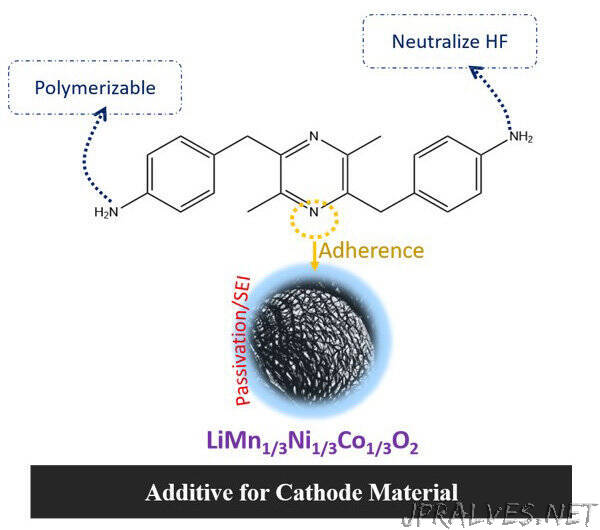
Recently, a team of researchers, led by Professor Noriyoshi Matsumi from Japan Advanced Institute of Science and Technology (JAIST), microbially synthesized 2,5-dimethyl-3,6-bis(4-aminobenzyl)pyrazine (DMBAP), a bio-based compound, as a potential additive for stabilizing the LiNi1/3Mn1/3Co1/3O2 cathodes. What sets their approach apart is the fact that, unlike existing additives, DMBAP is sustainable, eco-friendly, cost-effective, and non-toxic.
The team comprised Former Senior Lecturer Rajashekar Badam, Postdoctoral Research Fellow Agman Gupta, and Doctoral Course Student Noriyuki Takamori from JAIST, along with Professor Naoki Takaya, Assistant Professor Shunsuke Masuo, and Former Graduate Student Hajime Minakawa from the University of Tsukuba in Japan. Their findings have been published in the Scientific Reports journal.
“Although biomass-derived materials attract both researchers and the society in general, their applications in electric devices, including lithium-ion batteries, are still limited. This study focuses on novel microbial metabolites, particularly the unique pyrazine-derived diamine DMBAP from the gene cluster of Pseudomonas fluorescens SBW25, discovered in collaboration with Prof. Masuo. Its role as an electrolyte additive could impact the fields of sustainability and smart-cell industry,” explains Prof. Takaya, speaking of the motivation behind the study.
An initial theoretical evaluation revealed that the highest occupied molecular orbital (HOMO) of the DMBAP molecule was located at a higher position compared to a general-purpose electrolyte. This allowed it to be oxidized easily at the cathode surface and form a protective layer over it. In addition, the diamine in DMBAP prevented the dissolution of CEI.
The team additionally performed a detailed electrochemical evaluation of DMBAP for further analysis. The HOMO band energy was confirmed using linear sweep voltammetry, while X-ray photoelectron spectroscopy revealed C−N=C peaks indicative of oxidative electropolymerization. Cyclic voltammetry and charge-discharge studies showed that the DMBAP additive stabilized the LiNi1/3Mn1/3Co1/3O2 cathode by improving the battery’s rate capability, cyclic stability, coulombic efficiency, and capacity retention. Moreover, dynamic electrochemical impedance spectroscopy experiments demonstrated the formation of a low interfacial resistance CEI.
Based on these results, the team concluded that the DMBAP underwent sacrificial oxidative decomposition, forming an organic passivation armor on the cathode surface. This, in turn, restricted excessive electrolyte degradation and stabilized the structure of transition metal oxides on the cathode. In effect, this virtuous phenomenon increases the operating potential window of LiNi1/3Mn1/3Co1/3O2 cathode to 4.5 V versus Li/Li+. Moreover, the stabilizing effect of DMBAP on the battery system was remarkable for both half-cell and full-cell configurations.
“Microbially prepared pyrazine-amine compound DMBAP will boost the performance of lithium-ion secondary batteries essential for next-generation electric vehicles and drones. It will also promote the wider utilization of bio-based resources in the huge-scale automotive industry. Further, bio-based materials for energy storage devices will doubly reduce carbon dioxide emissions—during manufacturing and operation,” says Prof. Matsumi, discusses the future benefits of their work.
We certainly hope his visions come true, and soon!”
