“Planetary scientists prefer to call the thin gaseous envelope around the Moon as the ‘Lunar exosphere’ since it is so tenuous that the gas atoms very rarely collide with each other. While the Earth’s atmosphere near the mean sea level contains ~1019 atoms in a cubic centimetre of volume, the lunar exosphere contains ~ 104 to 106atoms in a cubic centimetre.
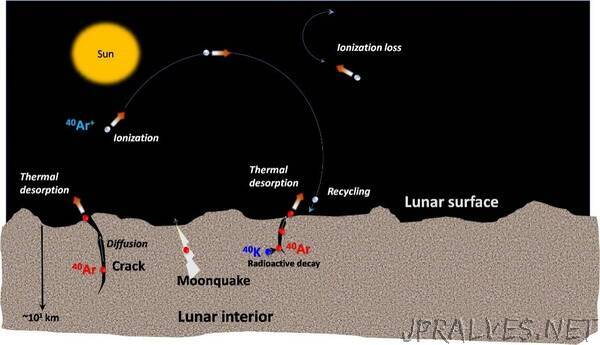
Argon-40 (40Ar), which is one of the isotopes of the noble gas Argon, is an important constituent of the lunar exosphere. It originates from the radioactive disintegration of Potassium-40 (40K), which has a half-life of ~1.2 X 109 years. The radioactive 40K nuclide, which is present deep below the lunar surface, disintegrates to 40Ar, which, in turn, diffuses through the intergranular space and makes way up to the lunar exosphere through seepages and faults.
The Chandra’s Atmospheric Composition Explorer-2 (CHACE-2) payload aboard the Chandrayaan-2 orbiter, is a neutral mass spectrometer-based payload which can detect constituents in the lunar neutral exosphere in the range of 1-300 amu (atomic mass unit). As part of its early operation, it has detected 40Ar in the lunar exosphere from an altitude of ~100 km, capturing the day-night variations of concentration. 40Ar being a condensable gas at the temperatures and pressures that prevail on the lunar surface, condenses during lunar night. After lunar dawn, the 40Ar starts getting released to the lunar exosphere (blue shaded region in figure).”
