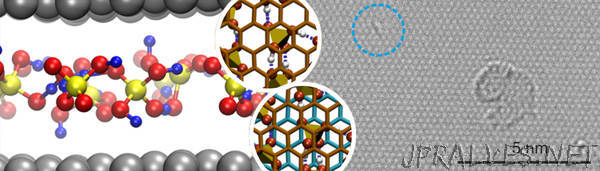
“Researchers from Freie Universität Berlin, Friedrich-Alexander-Universität Erlangen-Nürnberg (FAU) and Universität Ulm have made a significant contribution to the wet chemical synthesis of graphene from graphite, defining the mechanism on which it is based. They succeeded in solving the basic problem of how to separate an individual layer of graphene from a graphite crystal. The team led by Prof. Dr. Siegfried Eigler from Freie Universität Berlin was able to successfully stabilise individual layers of carbon from graphite using chemical functionalisation. By using computer simulations, the group led by Prof. Dr. Bernd Meyer from FAU was able to prove the mechanism. Dr. Ute Kaiser and her team at Universität Ulm succeeded in making the structure of the graphene manufactured using wet chemical methods visible at the atomic level with the help of electron beam microscopy. The results were published in the journal Nature Communications. An understanding of the chemical functionalisation of graphene and its synthesis is crucial if high quality graphene is to be made available in future. At present a lot of effort is being made to develop innovative electronics based on graphene rather than silicon (doi: 10.1038/s41467-018-03211-1).
Graphite is made up of various layers of graphene, which are in turn made of carbon. Carbon atoms in graphene are arranged in a honeycomb pattern, forming a two dimensional material with extraordinary electronic properties. Physical experiments on graphene were awarded the Nobel Prize in Physics in 2010.
The challenge faced when developing electronics based on graphene, however, is to obtain sufficiently large quantities of graphene, as it is virtually impossible to isolate graphene from graphite without damaging it. It is known that the space between the individual layers of graphene in graphite can be increased, but it is still difficult to separate the graphene layers in large quantities and stabilise the individual layers in solvent without destroying them in the process. Without stabilisation, individual layers of graphene would merge to form graphite or undefined carbon particles. The excellent properties of graphene would be lost as a result.
The researchers were able to prove that highly crystalline graphite with a well-defined order of layers is particularly well suited for being transferred to an intercalation compound in which molecules and ions are stored between the carbon layers. This is particularly successful if the layers of graphene are partially electronically oxidised, in other words positively charged. Molecule dynamic simulations show that the order of the graphene layers in the graphite together with electronic oxidation drastically reduce the friction of the molecules between the layers. Conversely, this means that a high level of friction of molecules in the layered material can restrict their movement, preventing graphite from being activated. Activation allows water molecules to react in the next step with the activated graphite. Alcohol groups are attached to the surface of the graphene, making it possible to separate individual layers of graphene and stabilise them in water. It was then possible to transfer this separated polar graphene to surfaces and reduce it to uncharged graphene. As a result, Prof. Dr. Ute Kaiser and her team from the Electron Microscopy Group of Materials Science at Universität Ulm were able to obtain an image of the structure of the high quality graphene at the atomic level.
The research was funded by the German Research Foundation within the context of the collaborative research centre ‘Synthetic Carbon Allotropes’ (CRC 953) and the individual project EI938/3-1. The team also received financial backing from the Graduate School Molecular Science (GSMS), the Fonds der Chemischen Industrie and the EU project Graphene Flagship.”
