“It´s a pity – most photocatalysts have an absorption maximum outside the sunlight spectrum. But now scientists from the e-conversion cluster were able to significantly increase the energy yield by using nanophotonic methods.
Photocatalysis allows to chemically store the energy from sunlight directly. One example is the splitting of water into oxygen and hydrogen – the most important renewable fuel needed for fuel cells. Although the principle of photocatalysis has been known for a long time, it has only been used to a very limited extent. The conversion from one form of energy to another is not efficient enough. One reason is the low optical absorption of the photocatalysts, because their materials are mostly transparent in the visible spectrum.
A team around the e-conversion members Professors Stefan Maier (LMU Munich), Emiliano Cortés (LMU) and Ian Sharp (Technical University of Munich, TUM) now demonstrated that the low absorption can be significantly increased using nanophotonic methods. Depending on the choice of catalyst material the increase is up to 150%.
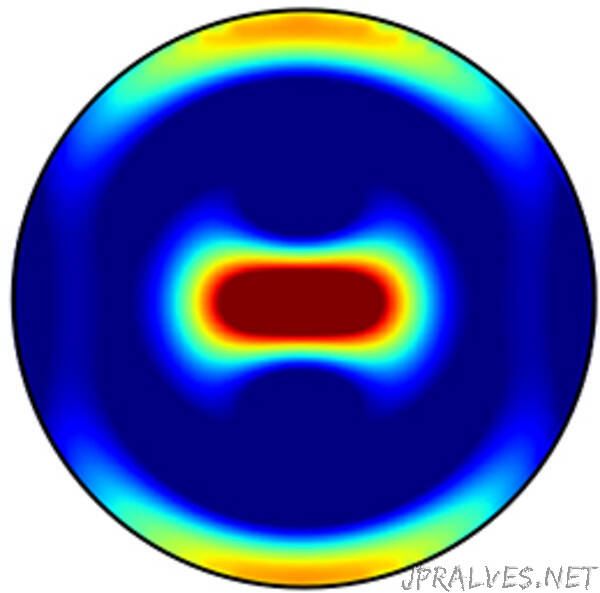
They formed sub-micrometer-sized cylindrical structures from a special variant of titanium dioxide, a classic material for photocatalysts. These so-called nanoresonators show a certain form of excitation in sunlight, the “Anapol”. The propagation of light in the nanoresonators overlaps in such a way that it concentrates inside and high electric fields are created. The desired consequence: The resonators show a significantly higher absorption in the sunlight spectrum.
The physicists have also managed to vary the spectral position of this enhancement mechanism in a controlled manner so that the concept can be applied to other materials. In the next step, the researchers will quantify the increased photocatalytic efficiency on scaled-up samples with several million of these nanoresonators.”
