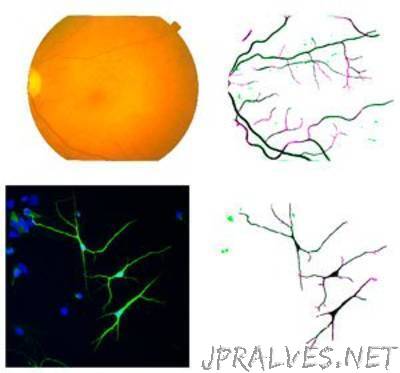
“A data-driven computational approach that recognizes filamentary sections of neurons and blood vessels may enhance medical diagnosis
A computational tool is now available for faster and more reliable screening and diagnosis of serious vascular and neurological conditions, including glaucoma, diabetic retinopathy, and Alzheimer’s disease, in their early stages. Developed by A*STAR researchers, the software automatically segments filamentary structures, such as retinal blood vessels and neurons, in various biomedical images.¹
Clinicians and biologists often need to evaluate the morphology of filaments in biomedical images to determine the presence and severity of vascular and neurological diseases. Filaments however can present a problem: as well as crossing and overlapping, they show variable diameters and degrees of twisting. This makes filamentary sections difficult to distinguish from backgrounds and, consequently, can skew visual assessments.
Several computer programs have been created to automate the segmentation process. These time-saving programs reduce human error, but their performance is typically optimized for specific tasks, which limits their use.
To widen the scope of these programs, Li Cheng and colleagues from the A*STAR Bioinformatics Institute, in collaboration with the Beijing Institute of Technology, have developed an algorithm that tackles segmentation as a general problem. “We have come up with a data-driven approach based on learning structured and contextual features,” says Cheng.
The researchers designed a two-step algorithm that sequentially extracts structured and contextual information from experimental data sets. During structured feature learning, the program captures informative image patches consisting of unique foreground textures that collectively help the machine segment them from backgrounds. This first step produces features that serve as input for the subsequent learning stage.
Next, contextual characteristics among patches are encoded into the algorithm using pre-existing machine learning tools, called boosted tree classifiers, to acquire more of the global information.
“We were very happy to find out that our algorithm can work across different tasks and performs well in general,” says Cheng. Compared to existing state-of-the-art methods, the algorithm delivered the best and almost best results over many different datasets (see image). Because of their specificity, most current methods typically work well for some tasks but not so well for others. “This is not the case for our algorithm,” adds Cheng.
According to Li Cheng, the new algorithm represents a stepping stone in his team’s long-term efforts to cure eye-related problems and decipher the structure and inner working of neuronal diagrams. His team is developing a series of tools to reconstruct, segment, and separate these elementary objects in imaging data. “We expect these tools to provide a solid platform for clinicians and biologists,” he adds.
The A*STAR-affiliated researchers contributing to this research are from the Bioinformatics Institute. For more information about the team’s research, please visit the Machine Learning For Bioimage Analysis webpage.”
