“Technion researchers have developed an innovative method for increasing the efficiency of electro-optical devices by increasing the interaction between light and matter. In findings published in Advanced Materials, the researchers report that they successfully implemented the method to produce a photoelectrochemical solar cell used to generate hydrogen from water, using solar energy.
The new method stems from the production of meta-materials that increase the light-matter interaction in ultrathin absorber layers. These materials are made of multi-layer structures that combine thin layers of insulators, metals and semiconducting metal-oxides.
The research was conducted by doctoral student Asaf Kay and postdoctoral researcher Dr. Barbara Scherrer with the guidance of the head of the research group, Prof. Avner Rothschild of the Department of Materials Science and Engineering.
Hydrogen is a promising fuel because it is the lightest and cleanest fuel. It can be produced by splitting water using solar energy in a process called photoelectrolysis, as demonstrated in this work. In this process, photons from the sunlight are absorbed by a semiconductor and the energy is transferred to the electrons in the material that absorbs the radiation. These electrons are used to break down the water molecules into hydrogen and oxygen (H2O → H2 + ½O2), which is done by breaking the chemical bond between hydrogen (H) and oxygen (O) in the water molecule (H2O).
The problem is that the electrons tend to quickly lose the energy received from the photons before they can break down the water. This phenomenon, called recombination, damages the efficiency of converting solar energy into hydrogen. Therefore, there is a clear need to increase the ratio between the rate of the photoelectrochemical reaction that splits the water and the recombination rate.
Photoelectrolysis is carried out by photoelectrodes that absorb the light and split the water. The photoelectrodes are required to absorb photons in as wide a spectrum as possible and to withstand adverse environmental conditions in corrosive solutions. In addition, it is important that they be produced from inexpensive, readily-available, and non-toxic materials. Due to the complexity of these challenges, no effective, stable, and inexpensive photoelectrodes were developed for many years.
In 2012, Prof. Rothschild and doctoral student Hen Dotan presented the development of iron oxide-based photoelectrodes, a major component of rust, in the journal Nature Materials. These photoelectrodes, which worked on trapping light in ultrathin iron oxide layers on specular back-reflectors, exhibited significant advantages over existing ones and broke the photocurrent record for iron oxide photoelectrodes.
But the method published in 2012 created new challenges related to the durability of the back-reflector – the specular layer beneath the iron oxide ultrathin layer. This back-reflector layer is made of aluminum or silver – materials that tend to oxidize or tarnish, respectively, and thus lose their efficiency in reflecting light back into the iron oxide layer. High temperature and oxygen atmosphere, conditions required for the production of the iron oxide layer, accelerate these processes and destroy that back-reflector layer. Consequently, Dr. Dotan developed a multi-layered device with various layers of protection that prevent the oxidation and tarnishing of the mirror. However, production has so far been very complicated.
In the present study, Asaf Kay and Dr. Barbara Scherrer developed an innovative manufacturing method that reverses the deposition order for producing the device. In this method, the iron oxide is deposited onto a piece of silicon before settling the metal layer (the mirror) above it. This process prevents the metal from oxidizing, and makes it possible to deposit it without the presence of oxygen and at a low temperature.
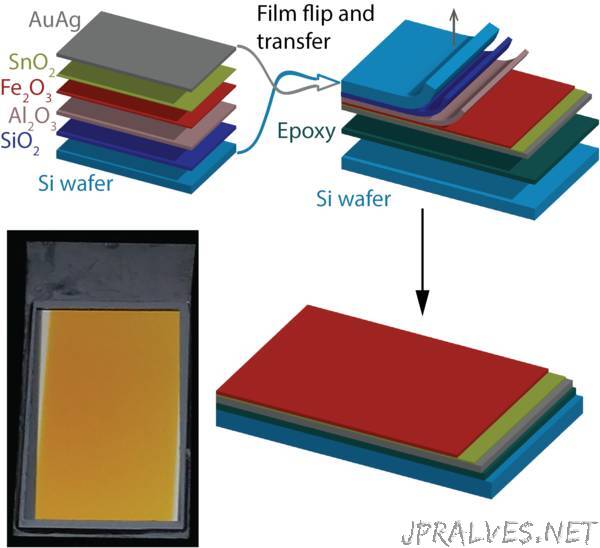
But the new method created a new problem: it placed the mirror above the iron oxide – in contrast to the structure required in the device. Therefore, the researchers added another production process in which they flip the entire structure, stick it upside down on another piece of silicon, and remove the first piece of silicon to expose the iron oxide. This process is illustrated in Figure 2.
The removal of the first piece of silicon was achieved with a combination of wet and dry etching processes used in microchip manufacturing for microelectronics. These processes require extreme caution so that the thin iron oxide, which is only about 10 nanometers thick, is not damaged. It is also important to avoid creating microscopic cracks in this process. To this end, a unique manufacturing process was developed at the Technion, using silica (SiO2) and alumina (Al2O3) protection layers, as shown in Figure 2. This process was developed by the professional team at the Sara and Moshe Zisapel Nanoelectronics Center and the Wolfson Microelectronics Center at the Technion.
The process was demonstrated by the production of a photoelectrochemical solar cell for splitting water, based on a thin layer of iron oxide (about 10 nanometers in thickness) on a mirror of silver and gold alloy. Beyond this specific example, presented in the Advanced Materials article, the new process is a breakthrough in the production and development of meta-materials that combine metal layers, semiconductors, and insulating materials to create electro-optical devices and innovative nanophotonic structures.
The research that led to the current breakthrough was funded by the European Research Foundation (ERC) under the FP7 program. The researchers thank the research funds that financed the work: ERC and the Marie Skłodowska-Curie fellowships which financed the employment of post-doctoral students Drs. Barbara Scherrer and Daniel Grave, the Sara and Moshe Zisapel Nanoelectronics Center, and the Wolfson Microelectronics Center, where the main processes in the development of the new production method took place.”
